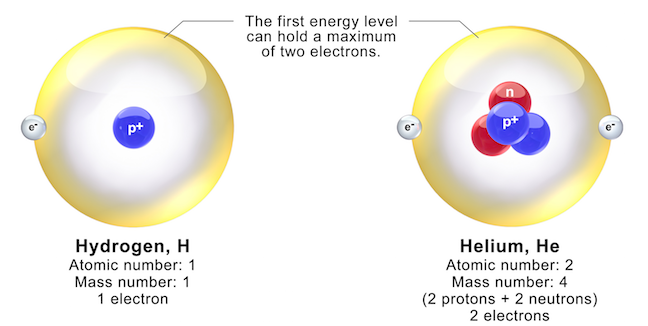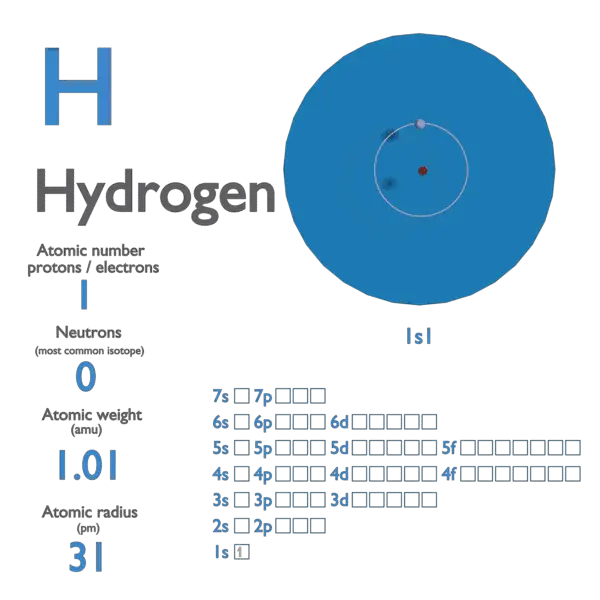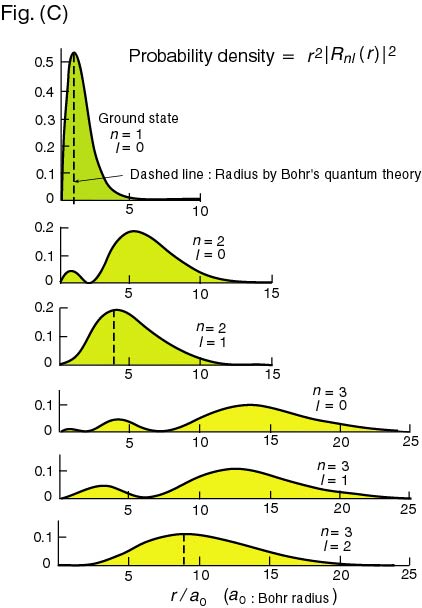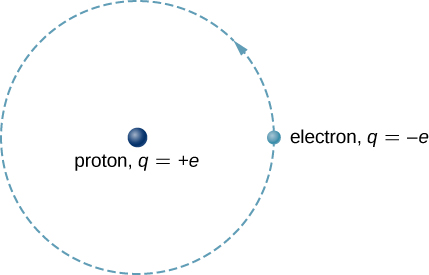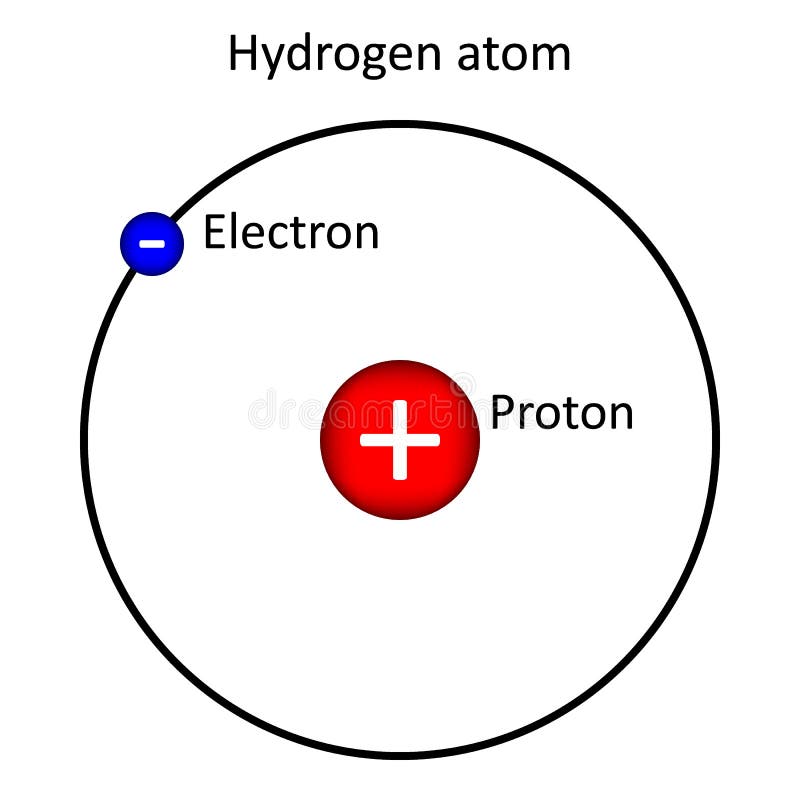
Hydrogen Atom on White Background. Stock Illustration - Illustration of electron, background: 121112400

Hydrogen spectrum | Line Spectra Of Hydrogen Atom | Atomic Structure & Periodic Properties | - YouTube

A) Schematic of Rydberg states in the H-atom (left) and in the H + H −... | Download Scientific Diagram

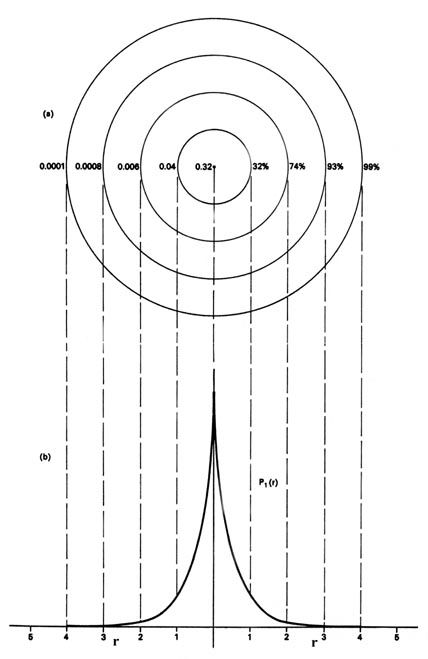
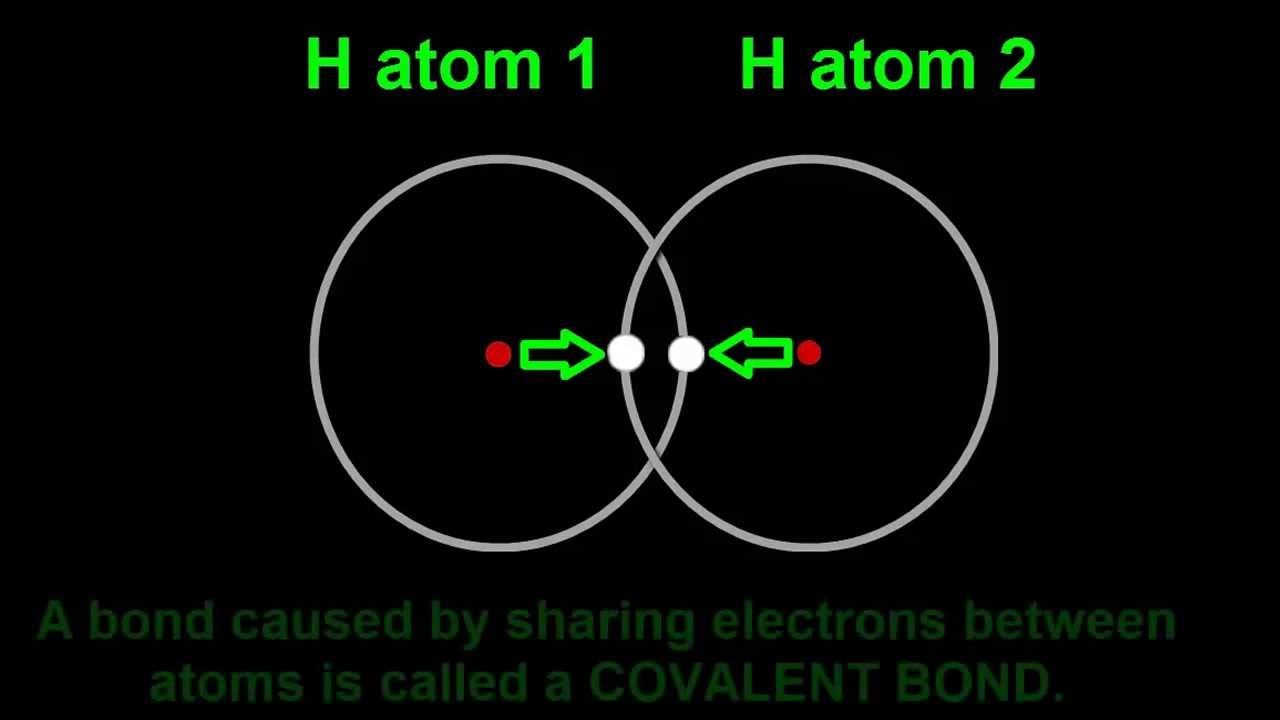
Energy levels of H atom are given by En = - 13.6/n^2 eV, where n is principal quantum number. Calculate the wavelength of electromagnetic radiation emitted by hydrogen atom resulting from the
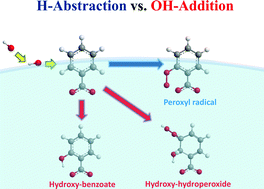
Extensive H-atom abstraction from benzoate by OH-radicals at the air–water interface - Physical Chemistry Chemical Physics (RSC Publishing)