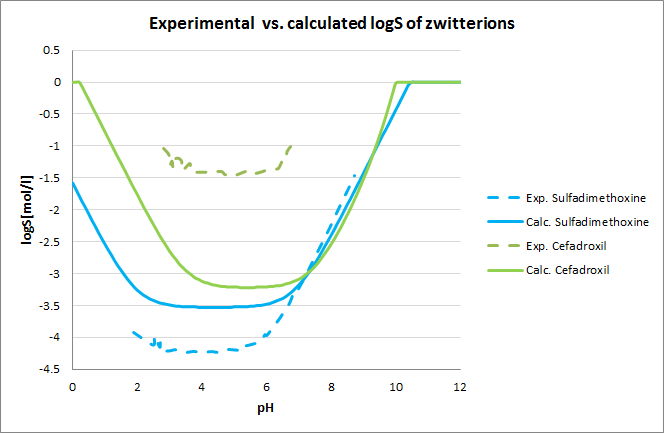
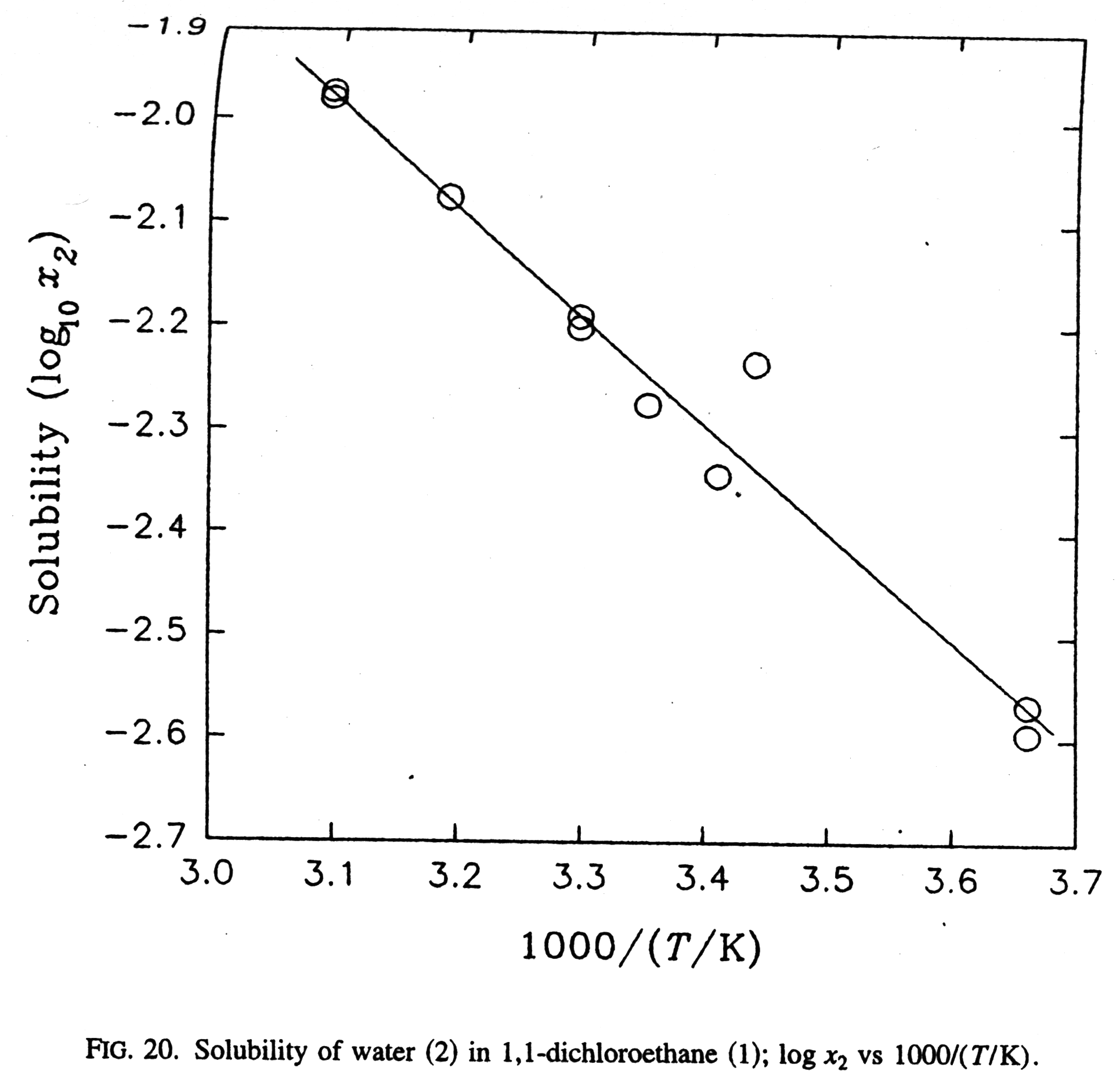
Development of Reliable Aqueous Solubility Models and Their Application in Druglike Analysis | Journal of Chemical Information and Modeling

Enhancement of water solubility of poorly water-soluble drugs by new biocompatible N-acetyl amino acid N-alkyl cholinium-based ionic liquids - ScienceDirect
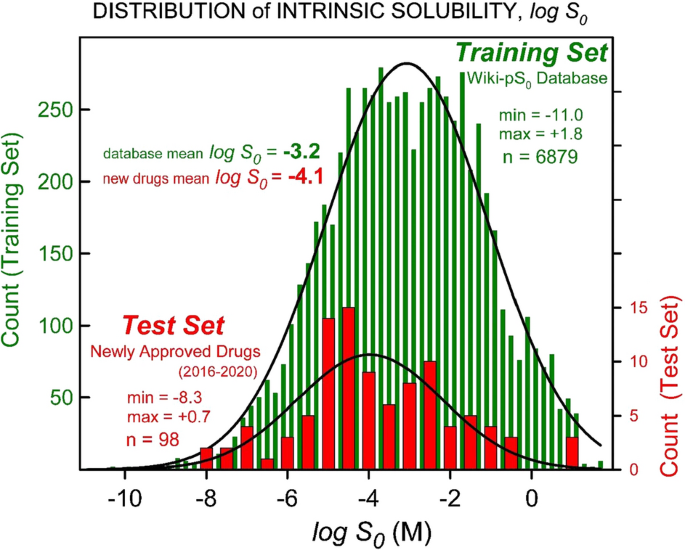
Predicting Solubility of Newly-Approved Drugs (2016–2020) with a Simple ABSOLV and GSE(Flexible-Acceptor) Consensus Model Outperforming Random Forest Regression | SpringerLink
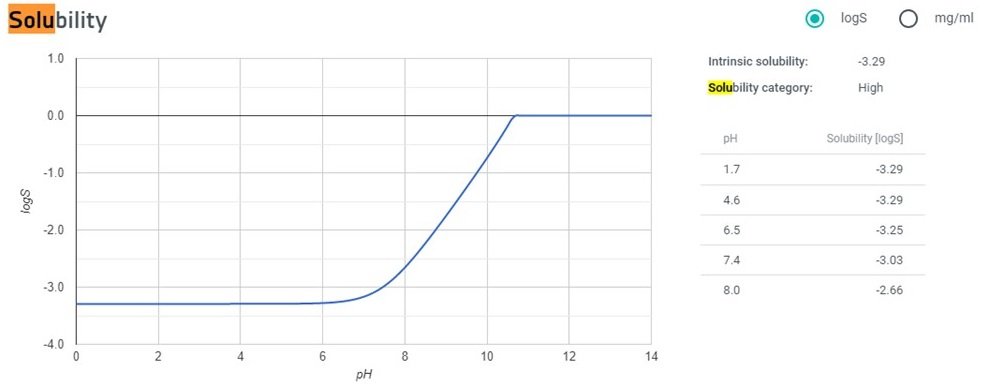
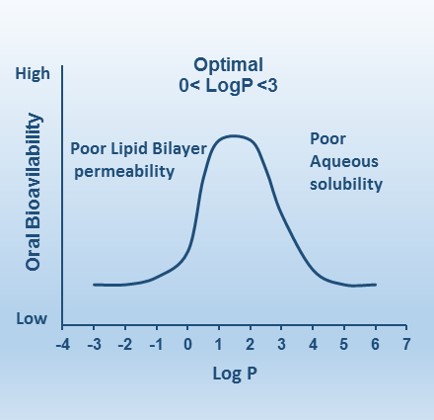
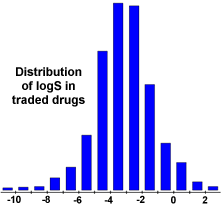
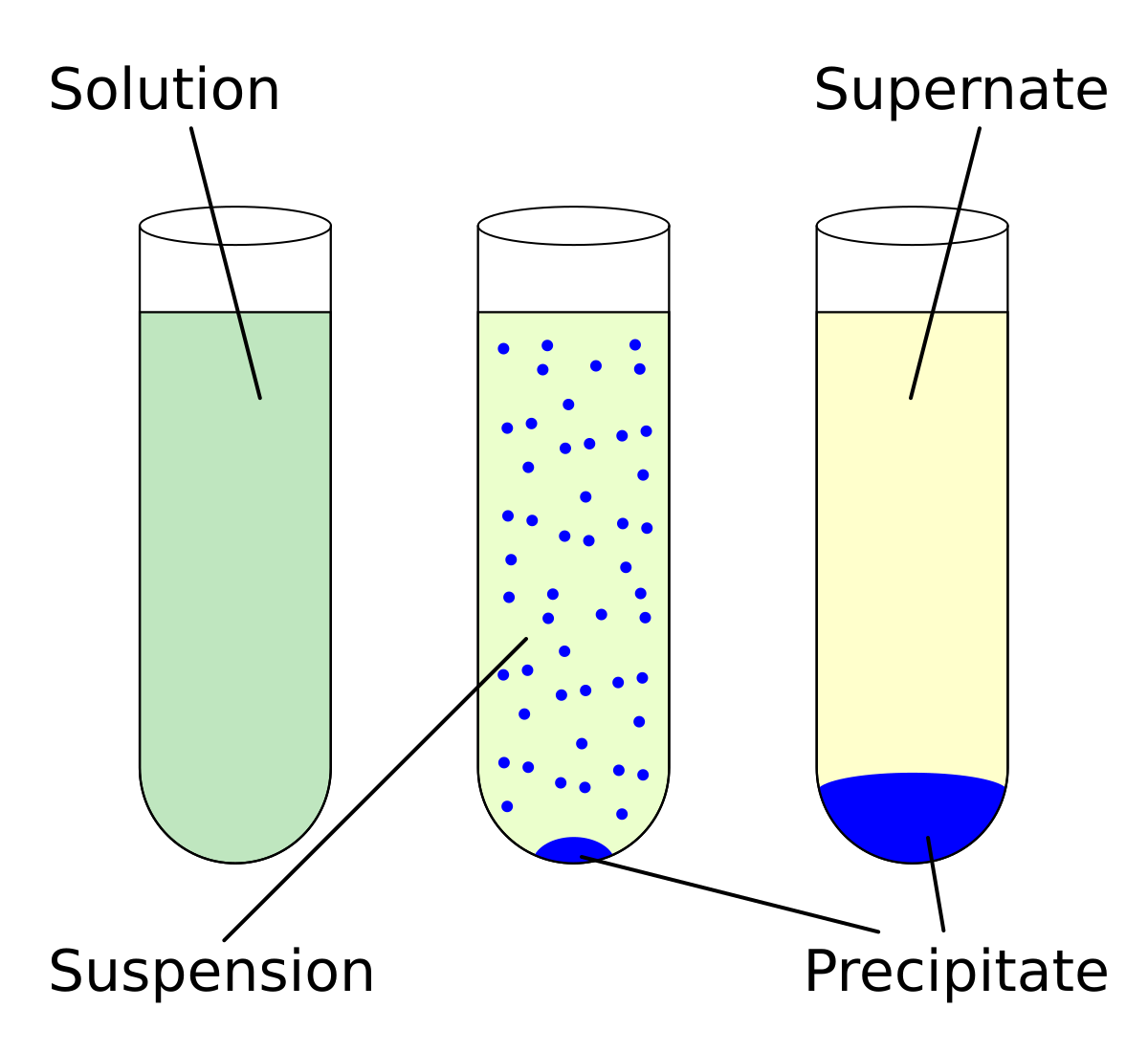
What is the difference between logP and logS regarding solubility in water? - Organic Chemistry - Science Forums

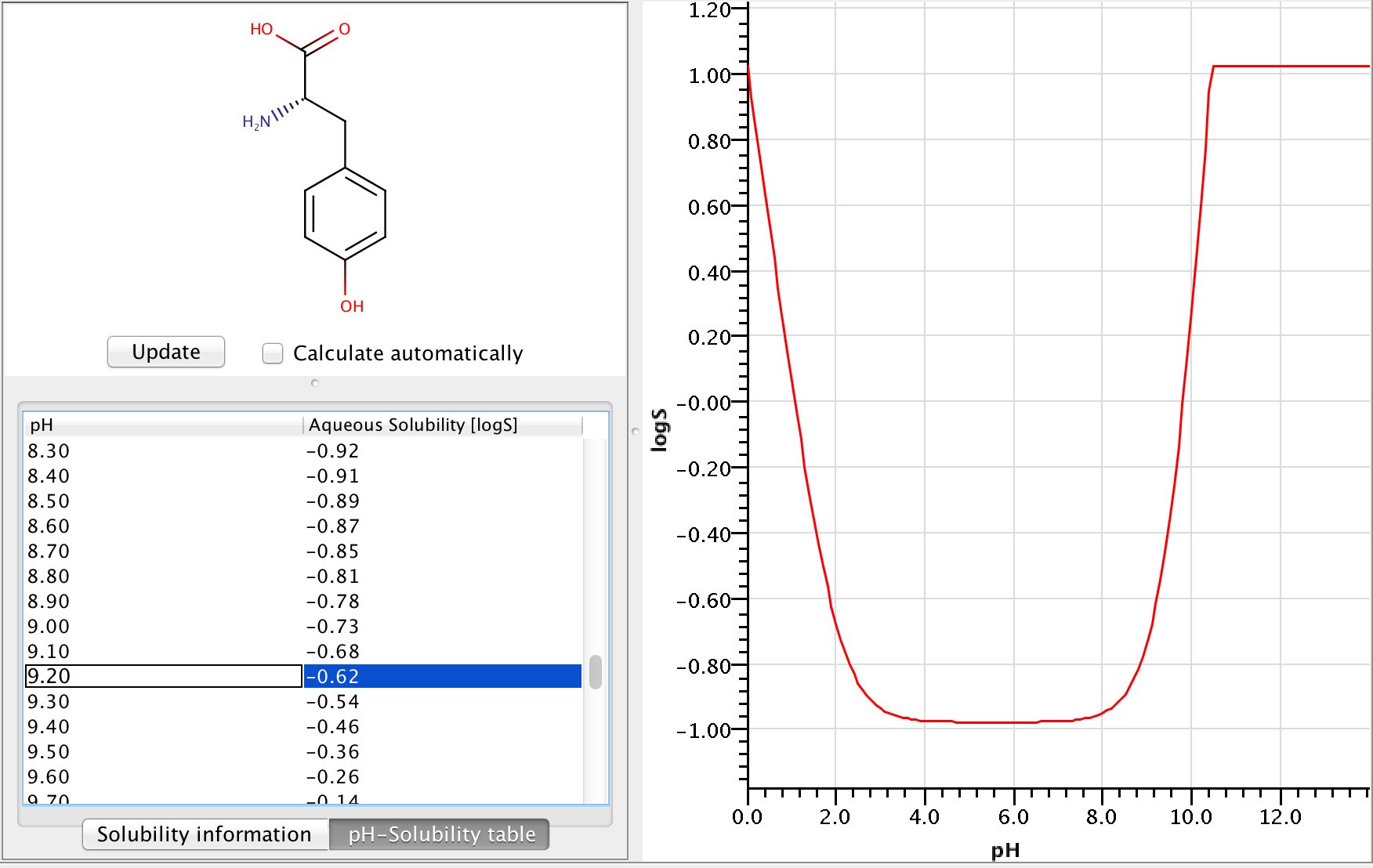
The solubility of a solute in water is dependent on temperature as given by: S = Ae^-ΔH/RT , where Δ H = heat of solution.Solute + H2O(l) Solution; Δ H = ±
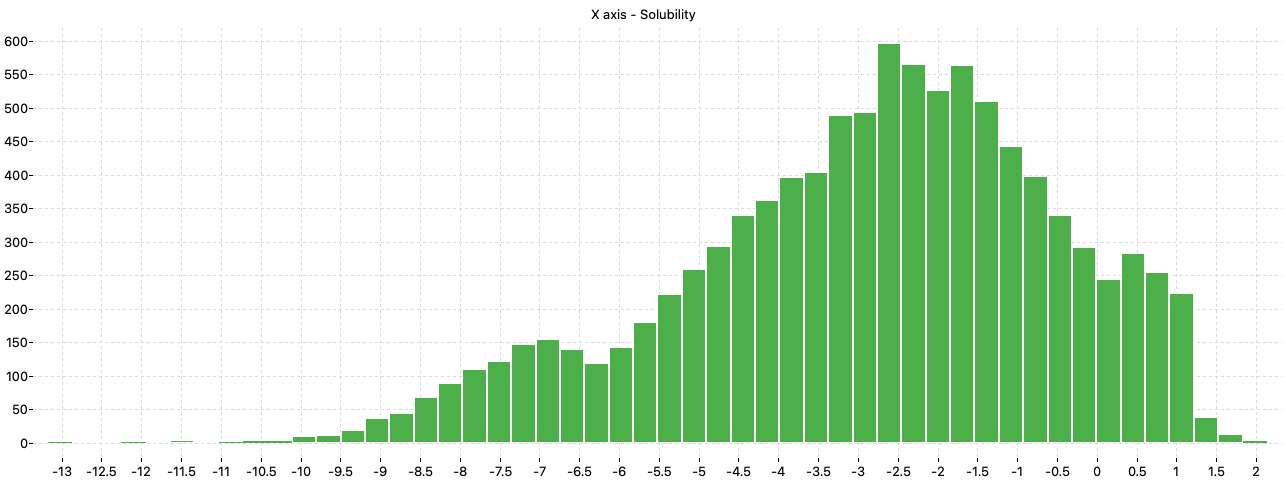
Predicting Aqueous Solubility of Organic Molecules Using Deep Learning Models with Varied Molecular Representations arXiv:2105.1

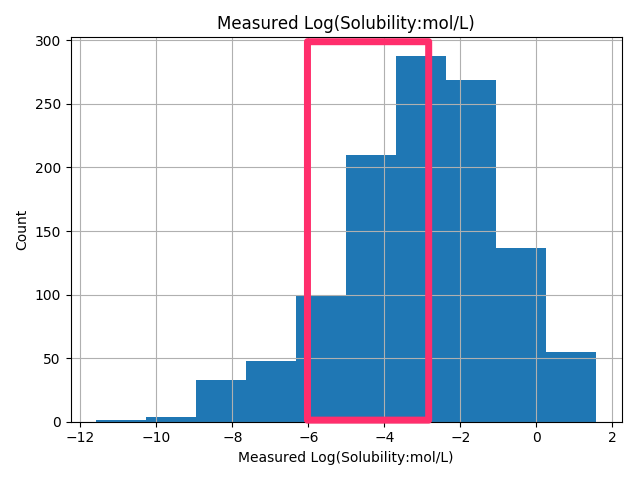
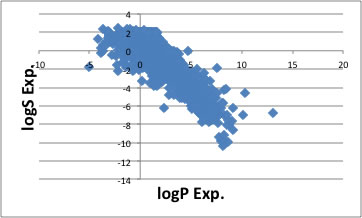
Studies of aqueous solubility (logS): (a) Clustering of the set of 1643... | Download Scientific Diagram